We explain the difference between polar and non-polar bonds in this chemistry tutorial. Then we will tell you what a polar molecule is, and what a non-polar molecule is. Last but not least, you discover what a dipole moment is.
Table of Contents
What do polar and non-polar bonds mean?
In simple terms, polar means oppositely charged while non-polar means equally charged. Covalent bonds can be polar or non-polar. Understanding electronegativity is essential to understanding polar and non-polar bonds.
What is electronegativity?
An atom’s electronegativity is a measurement of how much it wants to bond with another atom. On the periodic table, electronegativity increases from left to right and down each column. Using a scale from 0.7 to 4, the Pauling scale describes the electronegativity of an element. The element fluorine has the largest electronegativity, 4, indicating its electronegative nature. With an electronegativity of 0.7, cesium is the least electronegative element.
What makes a bond polar?
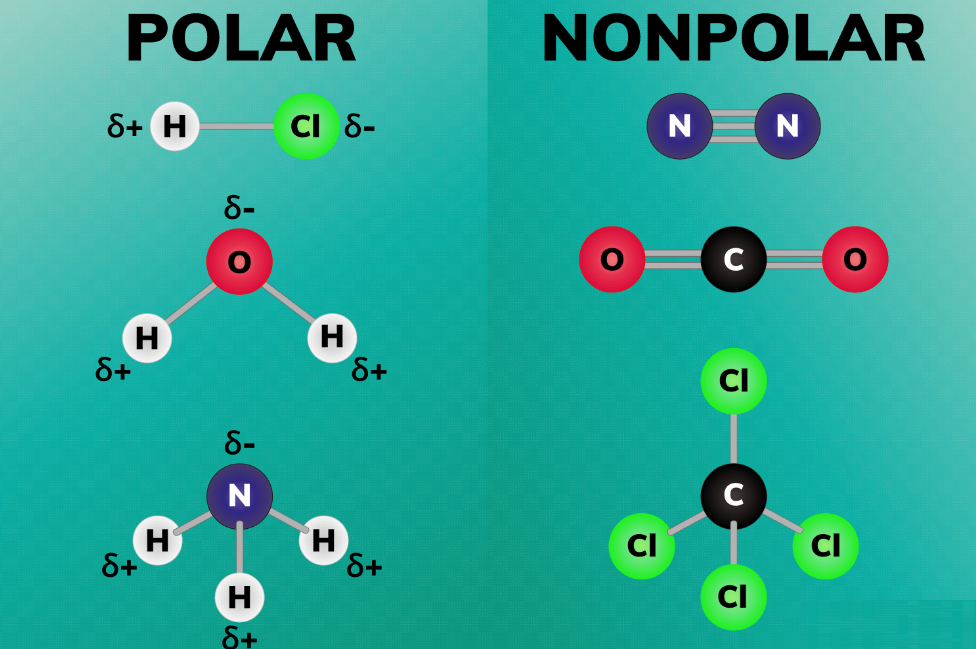
Polar bonds are a type of covalent bond. Polar bonds are formed when two or more atoms have significantly different electronegativities (>0.4). These bonds do not share electrons equally, which means the negative charge from electrons is not evenly distributed in the molecule. This causes a dipole moment. If there is one positive end of the bond and one negative end of the bond, then a dipole moment has occurred. Historically, polar bonds have been found in the bonds between hydrogen and oxygen in water. As a result of the electrodepositivity difference of 1, the bond is referred to as a polar bond. Hydrogen electrons are more attracted to oxygen electrons because oxygen is more electronegative.
What makes a bond non-polar?
Another type of covalent bond is the non-polar bond. This type of bond shares electrons equally, unlike polar bonds. A non-polar bond is one in which two or more atoms have the same electronegativity or electronegativities that are less than 0.4. For instance, the bond in chlorine is non-polar. Chlorine contains two chlorine atoms. It is equal sharing of electrons between the two atoms since there is no difference in electronegativity between them.
What are Polar Molecules?
In chemistry, the definition of a polar molecule is a molecule that has a charge on one side of the molecule that is not canceled out. It has a region of partial charge. It has one slightly positive end and one slightly negative end. The electrons are generally asymmetrical, with an uneven distribution.
Molecules with polar covalent bonds can have ionic or polar bonds. Molecules with two poles are called dipoles. The dipole moment is the result of measuring the amount of polarity in a molecule.
The electrons in a non-polar molecule can either be shared evenly, e.g. with a non-polar bond or symmetrically, as with carbon dioxide and carbon tetrachloride. Due to the symmetry, the molecules have dipoles, but due to the symmetry, they cancel each other out.
Polar molecules tend to stick together and line up in groups, which affects the properties of polar compounds such as water. When an electrostatic force is applied, water molecules will align themselves. Polar solvents tend to dissolve polar solutes, while non-polar solvents dissolve non-polar solutes.
Examples of Polar Molecules
Is HF polar?
Hydrofluoric acid HF is polar due to its large electronegativity difference between hydrogen and fluorine. The molecule is polar covalent.
Is water a polar molecule?
Definitely, since the molecule is bent and not symmetrical. A net charge results from electrons being attracted to oxygen atoms.
Is acetone polar or nonpolar?
The carbonyl group in acetone makes it somewhat polar. There are different degrees of polarity, and acetone is less polar than water because only part of the acetone molecule has a polar bond.
More examples of polar molecules
Sulfur dioxide SO2, ammonia NH3, carbon monoxide CO, ethanol C2H5OH, methanol CH3OH, hydrogen sulfide H2S, chloromethane CH3Cl, ozone O3, phosphorus trichloride (because it has trigonal pyramidal geometry) PCl3
Nonpolar Molecules
Nonpolar molecules are those in which the poles are neither positive nor negative. Charges are evenly distributed throughout the molecule. Nonpolar molecules are generally symmetrical, such as the tetrahedral carbon tetrachloride molecule. The trigonal planar structure of boron trifluoride is another example. When dipole charges cancel in symmetrical molecules.
When dissolved in nonpolar solvents, nonpolar molecules tend to dissolve well, but tend not to dissolve in water.
Examples of Non-polar Molecules
Is Carbon Dioxide polar?
No, CO2 is not polar, even though the bonds are polar. Due to the linear symmetry of the molecule, the negative charges around the oxygen atoms cancel each other out.
Is HCl polar or nonpolar?
Hydrochloric acid HCl has a polar bond due to chlorine’s electronegative property as compared to hydrogen’s. There is no symmetry that could cancel out the dipole charge.
More examples of non-polar molecules
Benzene C6H6, Methane CH4, Carbon Tetrachloride CCl4, boron trifluoride (because it is has trigonal planar geometry) BF3, hexane C6H14, nitrogen N2.
