By treating filter paper with any of the common pH indicators, you can make paper test strips to determine the pH of an aqueous solution. Litmus was one of the first indicators used for this purpose.
Basically, litmus paper is paper that has been treated with a mixture of 10 to 15 natural dyes derived from lichens (mainly Roccella tinctoria) that turns red in acidic conditions (pH 7). The dye turns purple when the pH is neutral (pH = 7).
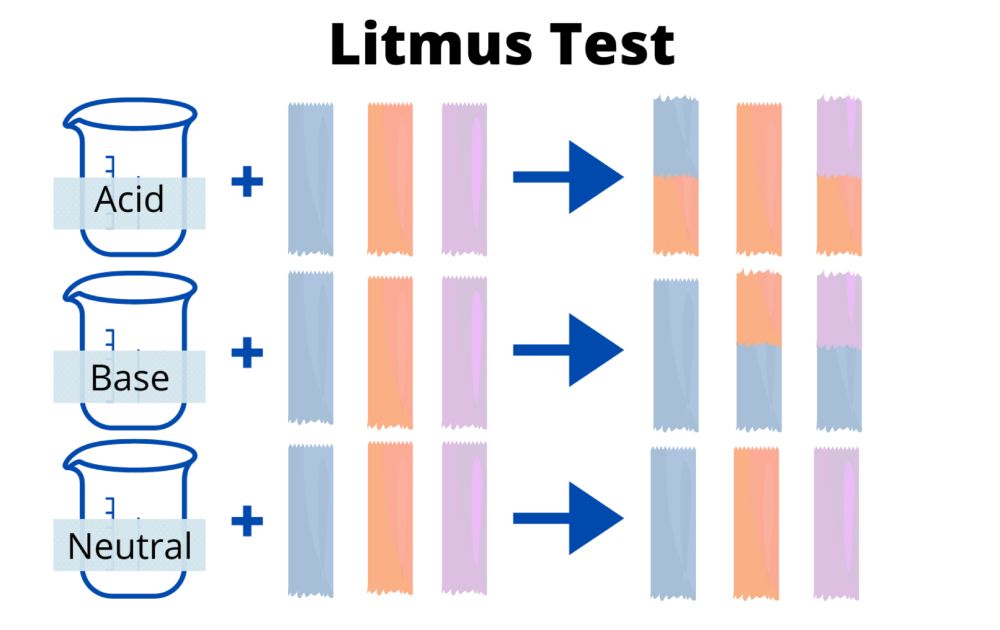
Table of Contents
History
Spanish alchemist Arnaldus de Villa Nova used litmus for the first time around 1300 CE. Since the 16th century, lichens have been used to extract blue dye. “Litmus” comes from the old Norse word for “dye” or “color.”
It is true that all litmus paper acts as pH paper, but the opposite is not true. Incorrectly referring to all pH paper as “litmus paper” is incorrect.
Fast Facts: Litmus Paper
- Using natural dyes from lichens, litmus paper is a type of pH paper.
- A small drop of sample is placed on colored paper to perform the litmus test.
- There are usually two colors of litmus paper: red and blue. When the pH is alkaline, red paper turns blue, and when the pH is acidic, blue paper turns red.
- It is most often used to test the pH of liquids, but it can also be used to test gases if dampened with distilled water before exposure.
Litmus Test
Place a drop of liquid sample on a small strip of paper or dip litmus paper in a small sample of the liquid. It’s best not to dip litmus paper in a whole container of a chemical – the dye might contaminate a valuable sample.
Litmus tests are used to determine whether a liquid or gaseous solution is acidic or basic (alkaline). Litmus paper or an aqueous solution containing litmus dye can be used for the test.
Litmus paper is initially either red or blue. In the pH range of 4.5 to 8.3, blue litmus paper changes to red, indicating acidity. Red litmus paper can indicate alkalinity with a change to blue when the pH is 8.3. Litmus paper is generally red below a pH of 4.5 and blue above a pH of 8.3.
When the paper turns purple, the pH is close to neutral. An acidic sample is indicated by red paper that does not change color. In the case of blue paper that does not change color, the sample is a base.
In non-aqueous liquids such as vegetable oil, pH paper won’t change color because acids and bases refer only to aqueous (water-based) solutions.
A gaseous sample can be tested with litmus paper dampened with distilled water. As the entire surface of the litmus strip is exposed to gases, the color of the litmus strip changes. The pH paper does not change color when exposed to neutral gases such as oxygen and nitrogen.
Reusable blue litmus paper can be made from red litmus paper. It is possible to reuse blue litmus paper that has changed to red.
Limitations
Litmus tests are quick and simple, but they have some limitations. It does not produce a numerical pH value, so it is not an accurate pH indicator. As a result, it gives a rough indication of whether a sample is acidic or basic. In addition to acid-base reactions, the paper can change colors for other reasons.
Blue litmus paper, for instance, turns white when exposed to chlorine gas. Rather than acidity or basicity, this color change is caused by bleaching of the dye by hypochlorite ions.
Alternatives to Litmus Paper
When you use an indicator with a narrower test range or a wider color range, you can get much more precise results than you would with litmus paper.
You are more likely to find cabbage at the local grocery store than lichen, for example, because red cabbage juice changes color according to pH from red (pH = 2) to blue (neutral pH) to greenish-yellow (pH = 12). Orcein and azolitmin produce results comparable to those of litmus paper.
